
ISO 9001 and ISO 13485 Standard have numerous likenesses, yet ISO 13485 contains necessities and terms that are exceptional to the clinical gadget industry. For organizations with certifications to more established older versions of ISO 13485 Standard, the deadline time to progress to the latest form was February 28, 2019.Įven though ISO 13485 Standard depends on ISO 9001 Standard, accomplishing a certificate doesn’t mean you are naturally agreeable with the other, and both require examining. The international organization for standardization (ISO) distributes the norm, and the ISO specialized council 210 dealt with the changes. The most recent adaptation of ISO 13485 Standard is ISO 13485:2016, which surpasses ISO 13485:2012, and was distributed on March 1, 2016. ISO 13485 additionally helps organizations that manufacture, introduce and manage medical devices with measure improvement, operational efficiency, and product improvement. establishing successful techniques for item traceability and review.complying with management and legal necessities.taking risk management to deal with item improvement.implementing a quality management system.The standard depends on ISO 9001 Certification yet contains extra prerequisites that relate explicitly specifically to manufacturing, installing, and servicing medical devices. and Canada) or 72 (outside US and Canada).ISO 13485 Standard is a quality management standard that is planned explicitly for the manufacturing of medical devices. Train staff on all of the requirements and areas of ISO 13485:2016Įmail or call 1-87 (U.S.
#WHAT IS ISO 13485 UPGRADE#
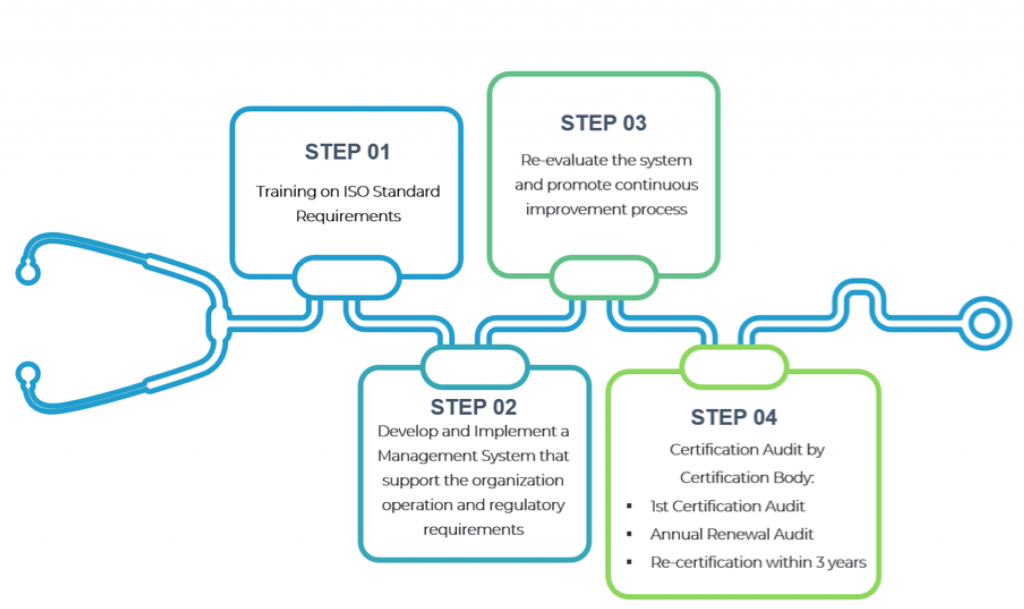
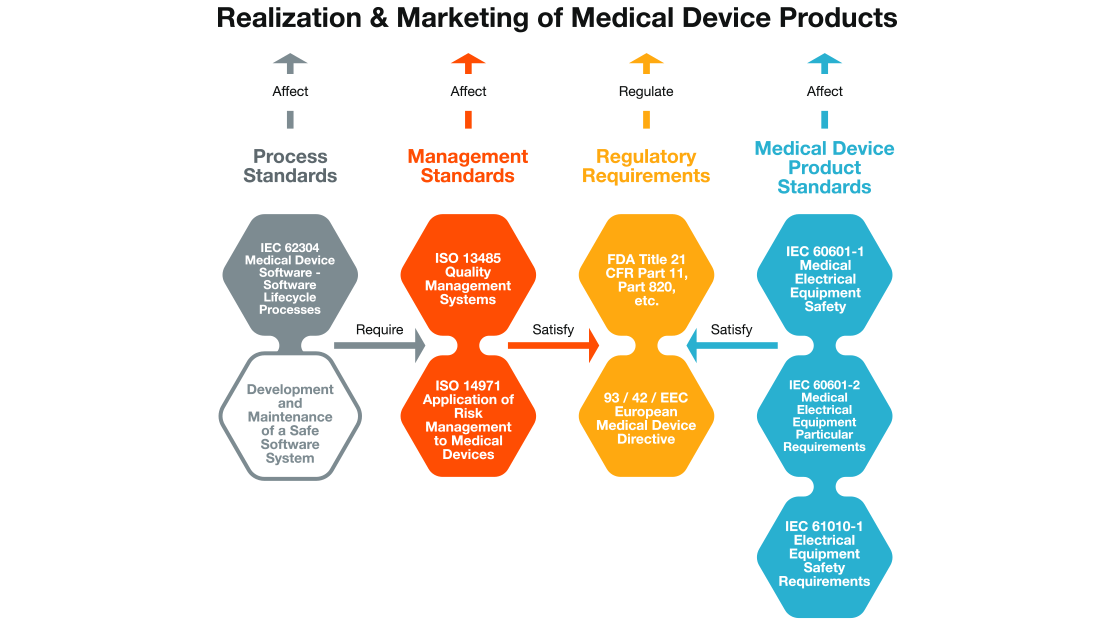

ISO 13485:2016 is an international standard that establishes the requirements for a quality management system specific to the medical devices industry.
